Frequently Asked Questions about GlazeMaster™
• Will GlazeMaster be available as an app for my iPad, smart phone, or iPod Touch? The short answer is 'no', there simply is not enough demand to justify the work to develop a fully functioning GlazeMaster app. Plus I know nothing about app programming. That said, it is pretty simple to put your recipes or batch mix sheets on your tablet in the form of pdf files.
Here are instructions that will do the trick for the iPad or other iOS devices like the iPhone or iPod Touch. If you have an Android device you will have to figure it out yourself, but it should be possible.
The first step is to "print" a pdf file instead of a paper copy of a recipe or batch mix sheet. This is easy as can be on a Mac; it takes a little more set-up work on a Windows machine. If you choose 'Print' on the main GlazeMaster menu, change to the recipe of interest, and then click 'Print Recipe', you will be taken to a printer dialogue box. On the Mac you then click on 'PDF' and choose "Save as PDF'.
On Windows you will probably need to install a PDF writer program which serves as a pseudo printer, i.e. it shows up as a choice in your print dialogue box, but it writes a pdf instead of putting ink on paper. There are several free-ware programs available and a recent review of them can be found at:
http://www.techsupportalert.com/best-free-pdf-writer.htm
You can keep those pdfs as a set of files or combine them into a single file using a program like Preview or pdfPen on the Mac. I'm sure there are similar programs for Windows. If you combine them consider making a title page so it will look its best on your tablet bookshelf.
So now you have a single pdf or a series of pdf files containing the recipes or batch mix sheets you want to have available on your tablet. iBooks can read pdfs, but the better, more capable, reader is the app GoodReader ($4.99 in the App Store). As an example of the flexibility of GoodReader, you can add or edit notes at specific spots on the pdf. These can be saved or not and, if saved, can be exported back to your computer or included if you email the pdf to another person. They cannot, however, be re-imported into your GlazeMaster file.
Whichever app you decide to use, you follow its standard procedure for loading the pdfs onto your tablet. The procedures are different for those 2 apps, though, so you will have to learn their specific procedure. However, neither are difficult.
I welcome hearing about other ways you have found to use a tablet, a smart phone, or an iPod Touch in conjunction with GlazeMaster. I will post those I think have broad applicability in the FAQs on the masteringglazes.com web site.
• May I use GlazeMaster screen shots on my web site or in my publication? Yes!! All we ask is a small credit along the lines of "Screen shots are from GlazeMaster™ glaze calculation software. Reproduced with permission of the author". It can be in small, but readable, type and only needs to appear once on your web site or in your publication. While not required, we would appreciate the word GlazeMaster being linked to http://www.masteringglazes.com/
Of course we also ask that you not use these screen shots to publish recipes from recently published books or journals without the recipe author's permission.
• I notice you don't list any limit formulas for earthenware glazes. Can GlazeMaster be used at firing temperatures other than Cones 6 and 10? Yes! GlazeMaster is useful at all firing temperatures.
Your question regarding limit formulas for earthenware glazes is a complex one, but I'll give it the old college try. First, no one that I am aware of, has done a study of limit formulas for earthenware temperatures to the depth Ron Roy and I have done it for cone 6. So what follows are some excerpts from the literature of what people have published for cones 06-04. Please remember they did not have glaze stability or durability as one of their criteria--only visually does it make "good glass". I will put some of my own thoughts at the end of this response.
Hamer and Hamer say that "acceptable limits" for leadless glazes in the range of 1832-2012 ºF would be
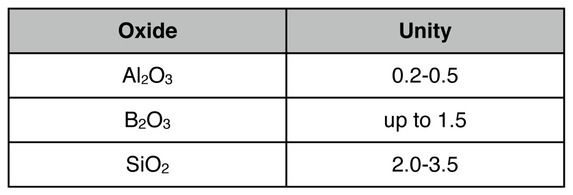
They don't give limits for alkali or alkaline earth fluxes.
David Green in his book "Pottery Glazes" gives "usual ratios" for cone 04 of
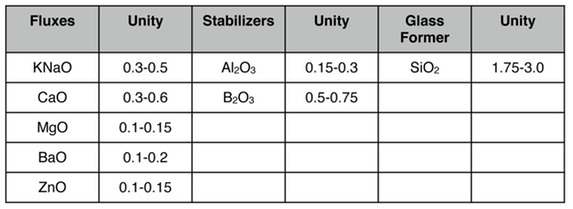
Zakin/Schmitz in the book "Ceramics: Mastering the Craft" give limit formulas for glossy, satin, and matte glazes. I'm only going to give the glossy ones below--you can find the others on pages 126-127 of that book. This is for cones 09-04 (quite a wide range).
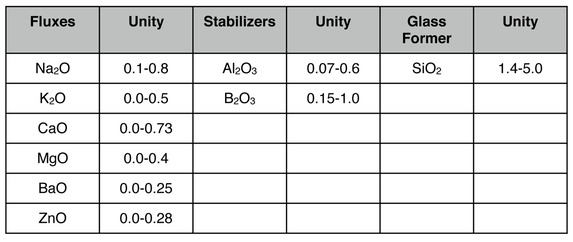
I think you can appreciate that these are so wide ranging as to not be very useful.
So what to do??? Though I have little data, I am firmly convinced that you have to have plenty of silica and alumina in any glaze if it is to be durable for use on functional pottery. After all, it is the composition of the cooled glaze that we are testing when we put pots in dishwashers and use them for acidic foods.
I also have a strong feeling that NO earthenware matte glazes are very durable --glossy ones can be. I just don't think you can get the composition right for durability AND for producing a matte surface without going up to cone 4 (or thereabouts) or above. If you are working with purely decorative surfaces you can, of course, make a full range of surfaces at earthenware temperatures.
So I would stick with the 4 rules Ron Roy and I developed in our work at cone 6 and suggest the following.
Silica should be at least 2.5, preferably 3 or above. You probably won't be able to get a good melt any above 3.5. Alumina should be at least 0.25, preferably above 0.3. Again you probably won't get a good melt above 0.4.
Boron should be fairly high. I don't think you will get decent melting below 0.5 and it is pretty unrealistic to push it much above 1.2 or so.
The other fluxes should lean toward the alkalis--you have to get every bit of fluxing power you can at earthenware temperatures. I would work from Green's set of limits with some modifications, e.g. while you must have some KNaO you don't have to have any MgO or BaO so those bottom limits would be zero.
Add up the above 4 paragraphs and here is what I would use.
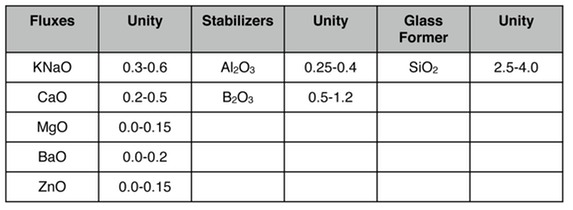
You might also get some benefit by incorporating some lithium (preferably sourced from spodumene rather than lithium carbonate) to both help with melting and help offset the higher expansion coefficient that results from a high level of KNaO and relatively modest levels of alumina and silica.
If you like you can enter the above as a new set of limit formulas for cones 06-04. Call it Hesselberth's Earthenware SWAG (the 'S' is for sophisticated) -- well, call it anything you like, but use it with great caution. Please remember that I am stretching my knowledge past its limit in the above; however this is where I would start. But then test, test, test. Be sure to do at least a vinegar test, particularly if you are wanting darker colors.
And if you do make use of the above limits, please let me know how it turns out.